
Coding and marking systems in the medical sector play a critical role in product safety, traceability, and regulatory compliance. From pharmaceuticals to medical devices, proper labeling of all products ensures patient safety while also providing significant operational advantages to manufacturers.
In this guide, we will examine the coding technologies used in the medical sector, UDI requirements, and how to choose the most suitable system in detail.
Why Are Coding and Marking Important in the Medical Industry?
There is zero margin for error in medical products. Therefore, each product must include the following information accurately:
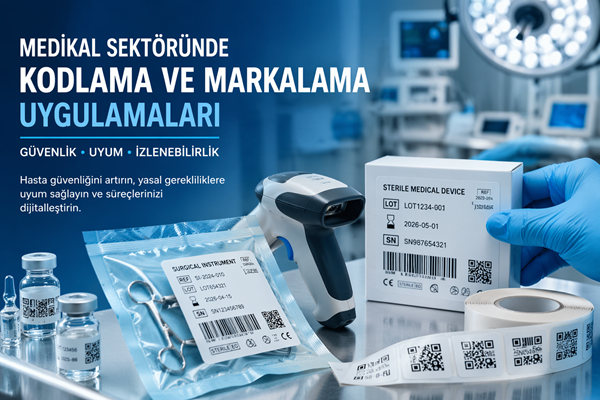
- Production date
- Expiration date
- Lot / batch number
- Serial number
- UDI (Unique Device Identification)
Key Benefits
- Product traceability
- Prevention of counterfeit products
- Efficient recall processes
- Compliance with international regulations (FDA, MDR)
Coding Technologies Used in the Medical Industry
1. Laser Marking
Laser marking is one of the most reliable and permanent solutions in the medical sector. It is especially preferred for surgical instruments, implants, and metal surfaces.
Advantages:
- Permanent and indelible marking
- Resistant to sterilization processes
- No chemicals required
- High precision
2. Thermal Inkjet (TIJ) Coding
TIJ printers are ideal for pharmaceutical boxes, blister packaging, and cardboard surfaces.
Advantages:
- High-resolution printing (QR codes and barcodes)
- Low maintenance cost
- Compact design
- Easy integration
3. Continuous Inkjet (CIJ) Coding
CIJ systems are used in high-speed production lines and offer the ability to print on various surfaces.
Advantages:
- Continuous operation
- Suitable for plastic, glass, and metal surfaces
- Fast-drying inks
4. Labeling Systems
Preferred especially in applications requiring variable data.
Advantages:
- Flexible data management
- Easy updates
- High readability
What Is UDI (Unique Device Identification)?
The UDI system ensures that each medical device is uniquely identified on a global scale.
UDI Includes:
- GTIN
- Serial number
- Production date
- Expiration date
Regulations:
- FDA (USA)
- MDR (European Union)
These regulations make coding systems mandatory for medical manufacturers.
How Should Proper Marking Be Done in Medical Products?
The following criteria should be considered when selecting the right system:
- Permanence (non-erasable)
- Readability (barcode/QR accuracy)
- Surface compatibility
- Hygiene and resistance to sterilization
Benefits of Coding and Marking Systems for Businesses
- Reduction in production errors
- Faster audit processes
- Increased brand reliability
- Digital traceability infrastructure
Conclusion
Coding and marking applications in the medical industry are not just a part of the production process but a fundamental element of patient safety.
With the right technology selection, you can both comply with regulatory requirements and make your production processes more efficient.

 EN
EN TR
TR